- News
Featured
FDA strengthens the role of analytical comparability in biosimilar development
- News
FDA strengthens the role of analytical comparability in biosimilar development
Analytical Comparability at the Center of Modern Biosimilar Development The U.S. Food and Drug Administration...
- News
FDA – Waives Clinical Efficacy Studies
A New Era for Biosimilars: Powered by Analytics In a historic regulatory shift, the U.S....
- Publications
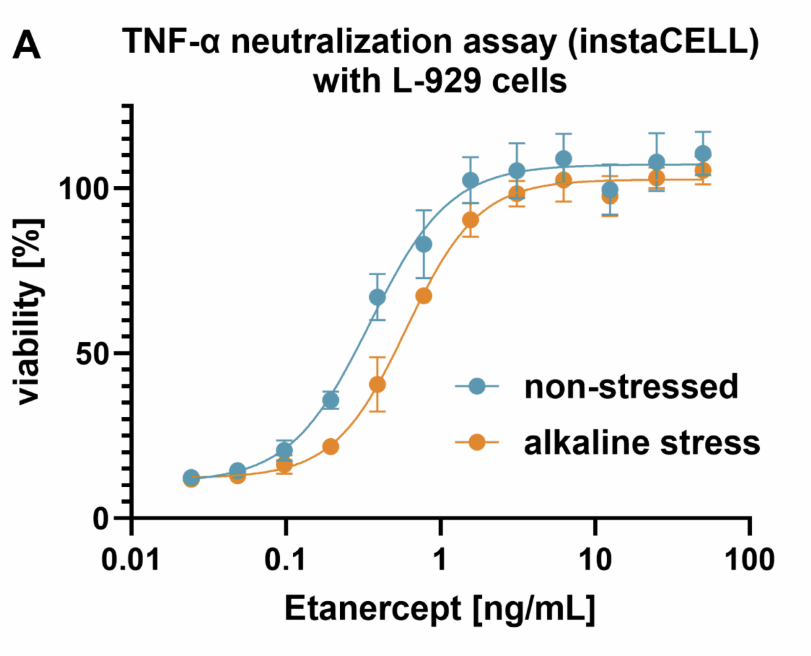
A holistic analysis platform for assessing protein modification and functional activity
Linking Protein Modifications to Functional Impact. Protein modifications are critical for the efficacy, safety, and...